Direct recycling technique uses electricity and low-cost chemicals to restore emerging lithium-iron-phosphate batteries. The only byproducts are water and oxygen.

Lithium-ion batteries enable cleaner energy and transportation systems, but their growing roles powering vehicles and balancing the grid presents a two-pronged problem: meeting the demand for critical minerals and managing mountains of waste.
It’s possible to recycle the components into new batteries, but most existing methods require a lot of energy and aren’t cost-effective for newer, cheaper battery chemistries.
Now a team of scientists at the University of Wisconsin–Madison has developed an economically viable and environmentally friendly technique to bring spent lithium ion batteries back to life.

“Battery recycling processes generally require large amounts of chemicals,” said Deok (DK) Roh, a postdoctoral researcher in Chemistry Professor Shannon Stahl’s lab who led the project. “This concept allows us to advance a much greener recycling process by replacing traditional chemical-heavy methods with electricity.”
The patent-pending technique uses electrochemistry to move positively charged lithium atoms (ions) into a solution that delivers them to the degraded battery material, restoring it to its original state. The process uses only electricity and an inexpensive alkaline solution; the only byproducts are water and oxygen.
“The approach is aligned with approaches that we have used for other applications,” said Stahl, an investigator with the Wisconsin Energy Institute. “DK recognized that electrochemical approach could be used for battery recycling. So he put those two pieces together.”
To test their results, Roh and Stahl collaborated with Eric Kazyak, an assistant professor of mechanical engineering who researches battery technologies. The results were published last month in Angewandte Chemie, the journal of the German Chemical Society.
“We designed a test that was intended to put the material through its paces,” Kazyak said. “Within a range of expected error the recycled material behaves essentially the same as the pristine.”
New batteries require new approach
The research, funded by the U.S. Department of Energy and the National Research Foundation of Korea, focused on an emerging battery chemistry that pairs lithium (Li) with iron (Fe) and phosphate (PO4), rather than more expensive nickel and cobalt.

These LFP batteries don’t hold quite as much energy per pound, but they are cheaper, safer, and last longer, making them a popular option for entry-level EVs. Last year, LFP accounted for over half of all electric vehicle batteries and more than 90% of grid storage, according to the International Energy Agency.
“That’s a massive load of waste that will eventually have to be disposed of,” Stahl said.
But the low cost makes recycling less economical.
Traditional recycling methods involve shredding the battery and melting it down, which requires a lot of energy, a process that Kazyak likens to “mining” the waste for the most valuable raw materials.
“You’re putting in a lot of energy to refine that back into something that you can use as a feedstock for your manufacturing,” Kazyak said. “Right now the nickel and the cobalt is where the value is, so that’s where most of the processes are focusing.”
Kazyak said the only viable path for LFP is direct recycling — reusing the same particles rather than breaking everything down into individual elements.
But those particles can be contaminated, fractured, or have defects.
“You have to find a way to deal with that,” Kazyak said. “That’s where the Stahl group’s innovation comes in.”

Putting the pieces together
Roh became interested in battery recycling while working in Korea, home to some of the world’s largest battery makers.
“I often think about how I can use my background in chemistry to solve pressing problems in our society,” Roh said. “Seeing the rapid growth of battery waste, I started looking into the specific bottlenecks in current recycling methods to see what exactly needed to be solved.”
He sought out Stahl because of his work in mediated electrochemistry, a method for delivering electrical charges to a hard-to-reach target using redox mediators – molecules that act as electron couriers.
“I realized we could combine the Stahl group’s expertise in mediated electrochemistry with my background in energy applications to design a new approach to this problem,” Roh said.
Over time, LFP loses lithium ions, creating holes in the crystal structure and causing the iron to oxidize. These structural changes “lock” the material and prevent it from holding a charge.
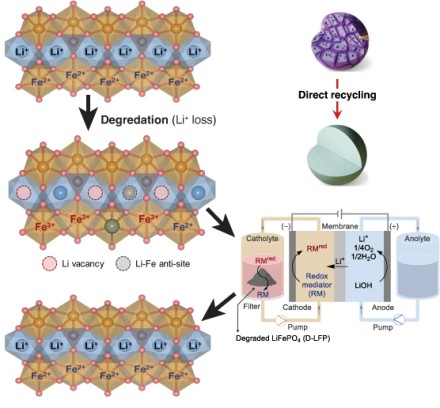
Previous direct recycling approaches require solid-solid contact with the cathode particles, which limits effectiveness. Others use organic solvents with redox mediators, introducing higher material and disposal costs.
Combining redox mediation and electrochemistry, Roh used electrical current to pull electrons from lithium hydroxide dissolved in water, forming oxygen, water, and positively charged lithium ions. The lithium ions pass through a membrane and recombine with the electrons in the mediator solution.
The charged mediator and lithium ions are then pumped through a reservoir, where they deliver the lithium ion and extra electron to the spent LFP. The mediator is then recirculated, and the process repeats.
“We don’t have to use an organic solvent,” Stahl said. “You’re using just water and lithium hydroxide, which is inexpensive.”
An evolving marketplace
A techno-economic analysis suggested the process could be economically viable, generating about $3 in profit for every kilogram of spent battery.
“I think there’s a pathway to larger scalability,” Kazyak said.
Stahl said the biggest challenge is not technology but timing.
“There’s just not a market for it right now because LFP is relatively cheap to make,” Stahl said. “But many of the new batteries coming online are going to be LFP materials. And so there’s going to be a recycling market coming. ”
The team is also working on ways to use their method in more real-world scenarios, where used batteries are ground up into a mix of materials known as “black mass,” and on other types of batteries.
“We’re not limiting ourselves to LFP,” Stahl said.





